Professor Catherine Hall

Understanding how limiting the brain’s energy supply causes disease
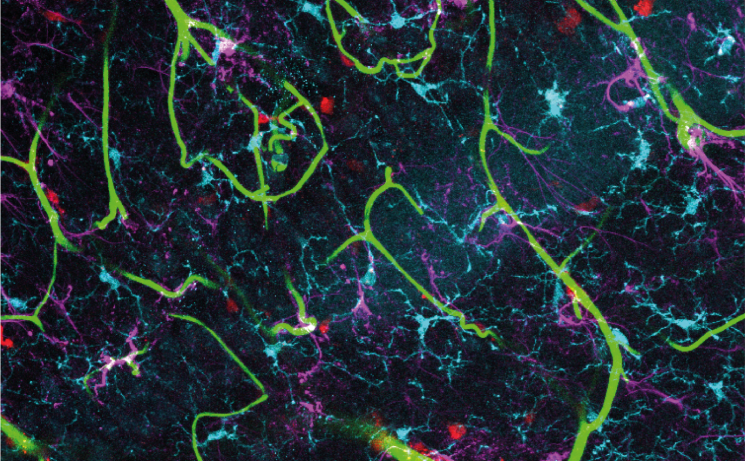
Brain activity is energetically expensive. To fuel regional brain activity, active neurons signal to dilate local blood vessels, increasing blood flow, seemingly matching energy supply with demand. Small decreases in brain blood flow happen in several diseases and are likely to be important drivers of pathology – for example, in Alzheimer’s disease, decreases in brain blood flow happen before symptoms develop and before the classic increases in beta amyloid and tau pathology occur.
In my lab we are trying to understand how these small changes in the brain’s energy supply can lead to disease. We use 2-photon imaging and optical measurement of haemodynamic responses in mouse cortex, corpus callosum and hippocampus, to measure neuronal, glial and vascular responses in vivo, as well as using biochemical assays, brain slice recordings and immunohistochemical labelling of fixed tissue. Together these measurements provide a rich understanding of how the brain changes at the onset of disease or when oxygen supply is reduced.
Several different and impactful new projects would be possible in this area, for example interrogating the impact of a reduced oxygen supply on different cell types in different brain regions, or testing whether improving oxygen delivery can protect brain function from decline in disease models.
Key references
- Shaw, K., Bell, L., Boyd, K., Grijseels, D.M., Clarke, D., Bonnar, O., Crombag, H.S. and Hall, C. N. (2021) Neurovascular coupling and oxygenation are decreased in hippocampus compared to neocortex because of microvascular differences. Nature Communications 12 (1), 1-16
- Clarke, Devin, Crombag, Hans S and Hall, Catherine N (2021) An open-source pipeline for analysing changes in microglial morphology. Open Biology, 11 (8). 210045 1-15. ISSN 2046-2441
- Grijseels, Dori M, Shaw, Kira, Barry, Caswell and Hall, Catherine N (2021) Choice of method of place cell classification determines the population of cells identified. PLoS Computational Biology, 17 (7). a1008835 1-32. ISSN 1553-734X
- Hall , C.N., Reynell, C., Gesslein, B., Hamilton, N.B., Mishra, A., Sutherland, B.A., O’Farrell, F., Buchan, A., Lauritzen, M. and Attwell, D. (2014) Capillary pericytes regulate cerebral blood flow in health and disease. Nature. 508(7494):55-60
- Hall, C.N., Klein-Flügge, M, Howarth, C and Attwell, D. (2012) Oxidative phosphorylation, not glycolysis, powers presynaptic and postsynaptic mechanisms underlying brain information processing. J. Neurosci. 32, 8940-51
- Hamilton, N., Attwell, D. and Hall, C.N. (2010) Pericyte-mediated regulation of capillary diameter: a component of neurovascular coupling in health and disease. Front. Neuroenerg. 2:5. doi:10.3389/fnene.2010.00005.
Visit Brain Energy Lab for a full list of publications and more details about Catherine Hall's lab.