Research and procedures involving animals
We are committed to meeting or exceeding all external ethical and regulatory requirements for research involving animals.
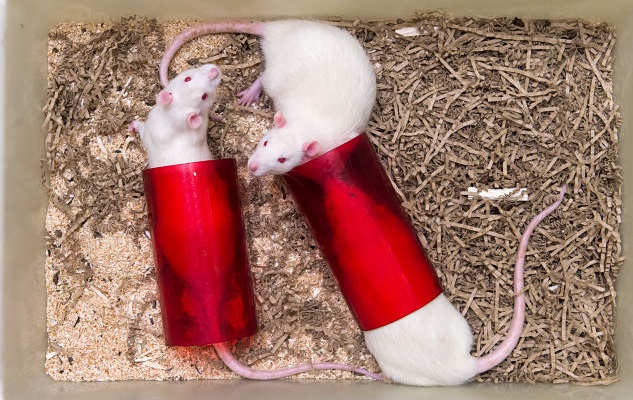
Rats housed in our animal facility, which operates to the highest standards of animal care and welfare. All research is only performed after animal welfare and ethical review has taken place.
Animal research at the University of Sussex
We are a signatory to the Concordat on Openness in Animal Research, which aims to help the public understand how and why animals are used on its behalf in scientific, medical and veterinary research in the UK. As part of the Concordat, we develop annual action plans to continue our commitment to openness and transparency.
Research using animals has made, and continues to make, a vital contribution to the understanding, treatment and cure of major human health problems; including cancer, heart disease, polio, diabetes and neurological diseases and disorders.
While new methods have enabled scientists and medical researchers to reduce studies involving animals, some work must continue for further fundamental advances to be made, but only after weighing the potential benefits of the research against the effects on the animals concerned.
The University's position is set out below:
- In the UK, research and teaching activities involving animals considered to be sentient and protected are governed by a range of legislation, including the Animals (Scientific Procedures) Act, 1986. Compliance of research involving these species is monitored by the Animal Welfare and Ethical Review Body (AWERB) and by the Home Office through its inspectors. In addition, the Animals in Science Regulation Unit (ASRU) regularly perform audits on our facilities and potential unannounced inspections.
- In addition to research performed on animals under the auspices of A(SP)A, we also perform ethical review on non-regulated teaching and research activities (non-A(SP)A).
- Ethical review at the University is performed by the Animal Welfare and Ethical Review Body with representatives from the research community, lay persons, the Named Veterinary Surgeon (NVS), Named Animal Care and Welfare Officer (NACWO), and the Named Training and Competency Officer (NTCO).
-
At the University of Sussex, we are committed to the highest standards of animal welfare and the principles of the 3R’s: Replacement, Reduction, and Refinement, to continually seek new approaches to improve research methods. These are:
-
Replace the use of animals with alternative techniques or avoid the use of animals altogether.
- Reduce the number of animals used to a minimum, to obtain information from fewer animals or more information from the same number of animals
- Refine the way experiments are carried out, to make sure animals suffer as little as possible. This includes better housing and improvements to procedures which minimise pain and suffering an/or improve animal welfare.
-
Animal statistics
The University of Sussex’s statistics on the use of animals in research are returned annually to the Home Office, and are laid out in the tables below.
| 2020 | 2021 | 2022 | 2023 | 2024 | |
|---|---|---|---|---|---|
| Mice | 6551 | 7715 | 5041 | 4717 | 4007 |
| Rats | 84 | 190 | 108 | 72 | 156 |
| Fish | 9380 | 11486 | 10550 | 9984 | 12944 |
| Frogs | 12 | 25 | 197 | 339 | |
| Other | 23 | 98 | 0 | 32 | |
| Totals | 16015 | 19426 | 15822 | 14970 | 17478 |
| Species | Numbers used (%) | ||||||
|---|---|---|---|---|---|---|---|
| 2020 | 2021 | 2022 | 2023 | 2024 | |||
| Mice | 40.9 | 39.7 | 31.9 | 31.5 | 22.9 | ||
| Zebrafish | 58.6 | 59.1 | 66.7 | 66.7 | 74 | ||
| Rats | 0.5 | 1.00 | 0.7 | 0.5 | 0.9 | ||
| Frogs | 0.1 | 0.2 | 1.3 | 1.9 | |||
| Other | 0.1 | 0.6 | 0 | 0.2 | |||
| Total | 100 | 100 | 99.97 | 100 | 100 | ||
| 2020 | 2021 | 2022 | 2023 | 2024 | |
|---|---|---|---|---|---|
| Sub Threshold | 4374 | 6349 | 5433 | 5078 | 11628 |
| Mild | 9301 | 9271 | 4311 | 3366 | 4140 |
| Moderate | 475 | 1408 | 1224 | 2663 | 1412 |
| Severe | 205 | 398 | 267 | 248 | 242 |
| Non-recovery | 1660 | 2000 | 4587 | 3615 | 56 |
| Totals | 16015 | 19426 | 15822 | 14970 | 17478 |
The University's Animal Welfare and Ethical Review Body review process
- The University's AWERB review process involves lay representation (including a representative from an animal welfare organisation) and other members both external and internal to the University. It provides ethical advice on standards of animal care, welfare and accommodation and ensures that those working with animals are aware of their responsibilities and receive appropriate training. Veterinary and animal care staff are actively involved in the ethical review of research, welfare and care of animals and provide ongoing advice and support to researchers where appropriate.
- In the case of animal research proposals, which are not covered by the Animals (Scientific Procedures) Act 1986, the Principle Investigator, or Supervisor (in the case of student projects) is still required to apply for approval through the University's review process.
AWERB meeting minutes (redacted)
AWERB draft minutes 18 March 2026 [DOCX 40.21KB]
AWERB draft minutes 14 January 2026 [DOCX 43.72KB]
AWERB draft minutes 9 September 2025 [DOCX 43.28KB]
AWERB draft minutes 4 June 2025 [DOCX 51.23KB]
AWERB draft minutes 19 March 2025 [DOCX 50.46KB]
AWERB draft minutes 14 January 2025 [DOCX 43.72KB]
AWERB draft minutes 8 January 2025 [DOCX 43.90KB]
AWERB draft minutes 17 September 2024 [DOCX 44.08KB]
AWERB draft minutes 14 June 2024 [DOCX 30.02KB]
AWERB minutes 30 April 2024 [PDF 1.81MB]
AWERB minutes 30 January 2024 [PDF 1MB]
AWERB minutes 21 April 2023 [PDF 1.57MB]
AWERB minutes 18 January 2023 [PDF 1.99MB]
AWERB minutes 7 December 2023 [PDF 3.44MB]
AWERB minutes 14 September 2022 [PDF 2.00MB]
AWERB minutes 15 July 2022 [PDF 4.20MB]
AWERB minutes 22 April 2022 [PDF 832.99KB]
AWERB minutes 27 January 2022 [PDF 454.84KB]
AWERB minutes 22 September 2021 [PDF 2.61MB]
AWERB minutes 8 July 2021 [PDF 2.94MB]
AWERB minutes 21 April 2021 [PDF 885.74KB]
AWERB minutes 12 Jan 2021 [PDF 959.01KB]
Contact us
Andy Cunningham
FIAT RAnTech
Biomedical Research Facility Manager
a.cunningham@sussex.ac.uk
+44 (0)1273 877010