Highly sensitive sensors successfully map electrical patterns of embryonic heart
By: James Hakner
Last updated: Wednesday, 11 November 2015
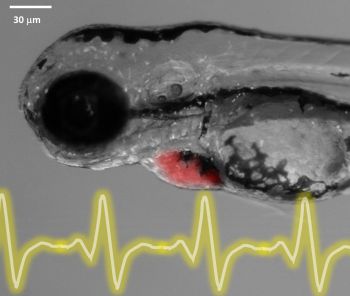
An example of the electrocardiogram mapped from the embryonic zebrafish heart three days after fertilization

Dr Elizabeth Rendon-Morales
Highly sensitive sensors have been successfully used to map the electrical activity of the developing heart in embryos, in a University of Sussex study published today (10 November 2015).
The study could lead to new insight into how heart rhythm abnormalities develop, the researchers say.
The team fine-tuned patented Electric Potential Sensing (EPS) technology, developed at the University, to detect the electrical signals of zebrafish embryos from just three days after fertilization. The findings have been published in the scientific journal Applied Physics Letters.
This is the first time that scientists have been able to see the full picture of the electrical activity in the developing heart of such a small organism, given that currently electrocardiography (ECGs) methods only measure the heartbeat.
Zebrafish hearts are remarkably similar to those of humans, making them a good model organism for scientific research. These latest results “push the frontiers” of the EPS sensor technology and could help scientists better understand how the heart develops – particularly during the early stages of life - and how defects originate.
Professor Robert Prance, head of the Sensor Technology Research Centre at the University of Sussex, said: “The EPS is a sensor technology that measures changes in an electric field in a similar way to a ‘perfect voltmeter’, drawing no current into the sample, making it non-invasive and safe to use. The sensor has already been used to carry out human ECG heart scans — or their cranial equivalent, electroencephalographs — saving patients from uncomfortable electrodes.
“Zebrafish embryo hearts are 2,500 times smaller than the human heart, so we needed to enhance the EPS design to make possible the detection of such cardiac activity.
“This could provide the biomedical research community with a novel instrumentation tool for performing cardio-electrophysiological studies at early developmental stages.”
Dr Elizabeth Rendon-Morales, a Research Fellow in the University's School of Engineering and Informatics and lead author of the study, said: “With this research we wanted to push the frontiers of the EPS sensor design towards its performance limits.
“Currently heart-related diseases such as congenital cardiac arrhythmias and long/short QT syndrome cost the EU economy almost 196 million euros a year. It is vital for us to develop tools that help our professionals understand as much as possible. This research could potentially generate new insights into the origins of such heart abnormalities.
“Using our technique based on electric field detection, we were able to obtain electrophysiological signals from living zebrafish embryos starting at three days post-fertilization. This is an excellent demonstration of the high sensitivity provided by the EPS sensor, which is also non-invasive and completely biocompatible.”
Although the technique has not yet been tested on human embryos, the study nevertheless provides a solid ‘proof of principle’ to underpin further research.
The team is now designing a multiplatform based on EPS arrays for the recording of electrophysiological signals simultaneously, which could potentially be used for applications in drug screening studies and neuroscience.
The EPS sensor was developed out of research led by Professor Prance, in collaboration with the University’s business incubation network Sussex Innovation. A licensing agreement was signed in 2010 with Plessey Semiconductors to bring new applications to market and it was shortlisted in the 2011 Times Higher Education Awards for technological innovation of the year.
Notes for editors
University of Sussex Media Relations team: James Hakner (01273 877966) and Jacqui Bealing (01273 877437) – press@sussex.ac.uk.
Non-invasive electrocardiogram detection of in vivo zebrafish embryos using electric potential sensors is available online at http://scitation.aip.org/content/aip/journal/apl/107/19/10.1063/1.4935249.
Images:
Journalists are free to reproduce the two images. Please contact press@sussex.ac.uk for the files and credit Dr Elizabeth Rendon-Morales.
Further information: http://scitation.aip.org/content/aip/journal/apl/107/19/10.1063/1.4935249